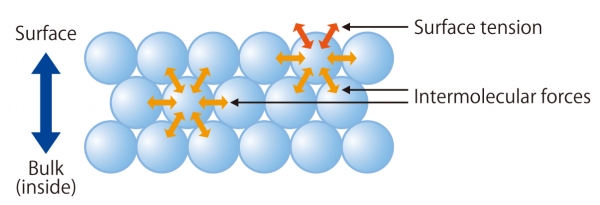
What is Surface Tension?
Intermolecular forces work between the molecules and the molecules of the liquid to try to minimize the surface area of the liquid. If Surface Tension is this force, the magnitude of the force that is exerted is a significant phenomenon, such as Wettability.
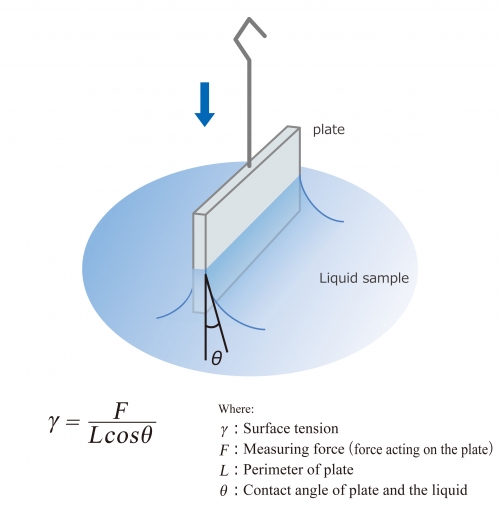
Wilhelmy Plate Method
When the measuring unit (Wilhelmy plate) contacts the liquid surface, the liquid will wet the plate upward. In this case, surface tension acts along the perimeter of the plate, and the liquid pulls on it. This method detects the pulling force and determines the surface tension.
Du Noüy Ring Method
The Noüy ring is made of platinum to ensure a contact angle of zero degrees with liquids. The method has a longer history than other modern measurement methods, dating back to its establishment, and has been adopted by some industrial standards.
First, a ring parallel to the liquid surface is submerged. Then, the ring is gradually pulled vertically away from the surface. In this process, the surface tension of the liquid film attached to the ring generates a force on the ring. This force changes as the ring is drawn farther. Using the maximum value of this force, surface tension is determined by the following formula:

C in the formula is a correction factor and a constant that depends on the ring size and the liquid sample density. This factor corrects the effects of the tension direction and the liquid film's shape. Our DyneMaster series automatically corrects them using the Zuidema-Waters formula.
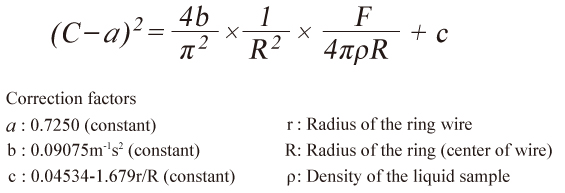
Lamella Length Measurement
One feature of surface tension measurements using the du Noüy tear-off method is that it also provides lamella length values. The lamella length indicates how long a liquid film can be prolonged before it detaches from the ring.
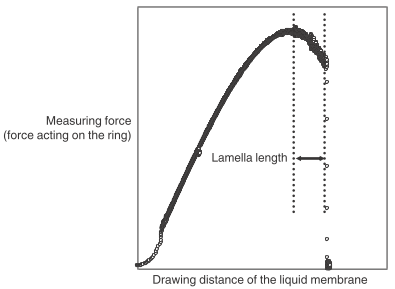
Drawing up the liquid film at high speed negatively influences its lamella length because it can cause the film to detach from the ring before reaching its full length. Therefore, a low constant speed and smooth movement of the sample stage are prerequisites for accurate measurements and good repeatability.
The lamella's length provides information on the onset of curtain breakup in curtain coating, foam stability, and antifoaming properties. It also indicates the ability of paints and coating liquids to pick up onto coating rolls.
Related Products
Pendant Drop Method
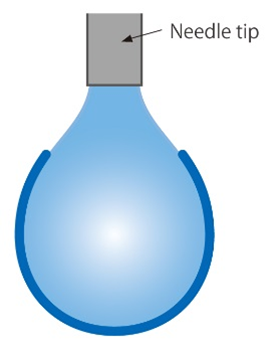
A liquid droplet suspended from a needle tip forms the characteristic shape of a pendant. It can be suspended in a gas phase or in another liquid. The shape of the pendant drop depends on the liquid volume (weight), the density difference Δρ between the phases, and the surface or interfacial tension. Surface or interfacial tension can be determined by taking a photo and analyzing the droplet’s contour.
The pendant drop method is suitable for measurements of highly viscous liquids, molten polymers, waxes, and solders.
Compared to other measurement methods, such as the Wilhelmy plate or du Noüy ring, the pendant drop method has advantages in the following points compared to the conventional Wilhelmy plate and du Noüy ring method:
- ✔ Volume
Measurements only need a few uL of liquid
- ✔ Temperature
Temperature can be controlled up to 400℃, enabling measurements of waxes and molten polymers
- ✔ Time
Due to its short-term exposure to air, it is suitable for liquids with rapidly changing surface tension
Young-Laplace equation
The Young-Laplace equation uses the density difference between the phases; a calculated curve according to the Young-Laplace equation will be fitted onto the droplet’s contour.
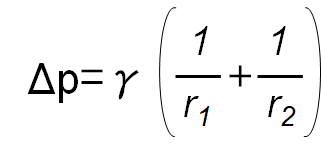
γ: Interfacial tension
Δρ: Density difference
r1, r2: Radii of curvature
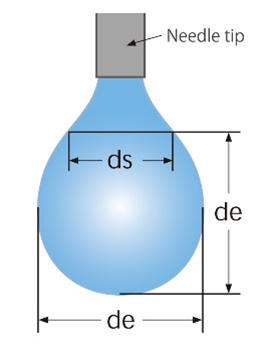
ds/de equation
The ds/de equation uses the interfacial tension, the equatorial diameter de, and the diameter ds, measured at the distance of de from the bottom of the droplet, to determine the surface tension.
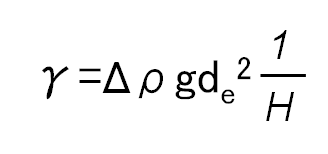
γ: Interfacial tension
Δρ: Density difference
g: Gravity
de: Equatorial diameter of the droplet
1/H: Correction factor derived from ds/de
Related Products
Dynamic Surface Tension
Surfactants lower surface or interfacial tension over time by adsorbing to the newly formed surface or interface. During the time from a freshly created surface to the equilibrium value of surface tension, dynamic surface tension can be measured; at the liquid's equilibrium state, static surface tension can be measured. Detergents and coating solutions are used in processes that continually create new surfaces or interfaces. Thus, determining the Dynamic Surface Tension is highly important.
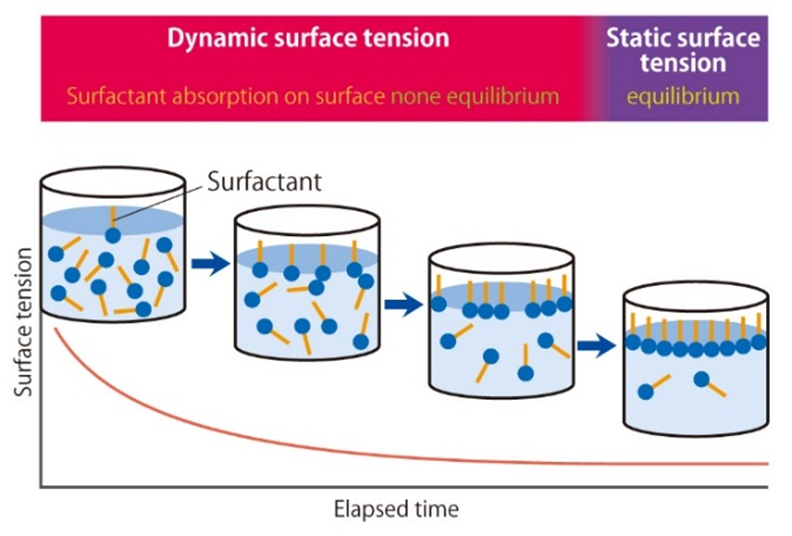
Maximum Bubble Pressure Method
The Maximum Bubble Pressure Method (MBPM) is the industry standard for determining dynamic surface tension over a broad timescale — from long-term equilibrium down to milliseconds. By analyzing the peak pressure required to form a gas bubble through a high-precision capillary immersed in a liquid, this method reveals the rapid, time-dependent adsorption kinetics of surfactant molecules before they reach thermodynamic equilibrium.
This technique is critical for optimizing high-speed industrial processes such as inkjet printing, industrial coating, detergent formulation, and spraying applications, where fresh interfaces are created in fractions of a second.
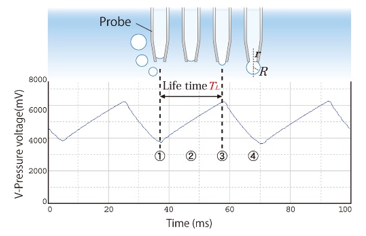
When pressurized air flows continuously through a capillary, the pressure within it oscillates in a precise, regular cycle from stages ① to ④:
Bubble Lifetime / Surface Age → ① to ③
The interval from initial bubble growth ① up to the peak pressure point ③ is defined as the lifetime (or surface age). This is the critical window during which surfactant molecules migrate and adsorb to the freshly created gas-liquid interface.
The bubble pressure reaches its absolute maximum ③ at the exact moment the bubble forms a perfect hemisphere, meaning its radius of curvature ( R ) equals the radius ( r ) of the capillary outlet.
Dead Time → (3) to ④
Once past the hemispherical peak ③, the bubble becomes unstable and expands rapidly as the pressure drops sharply, continuing until it finally detaches from the capillary tip ④. This rapid growth and release phase is known as the dead time, during which no meaningful surfactant adsorption occurs.
Cycle Reset → ④ back to ①
Immediately following detachment, the pressure drops to its baseline, a new bubble begins to emerge at the capillary tip, and a new measurement cycle commences from ① to ④.
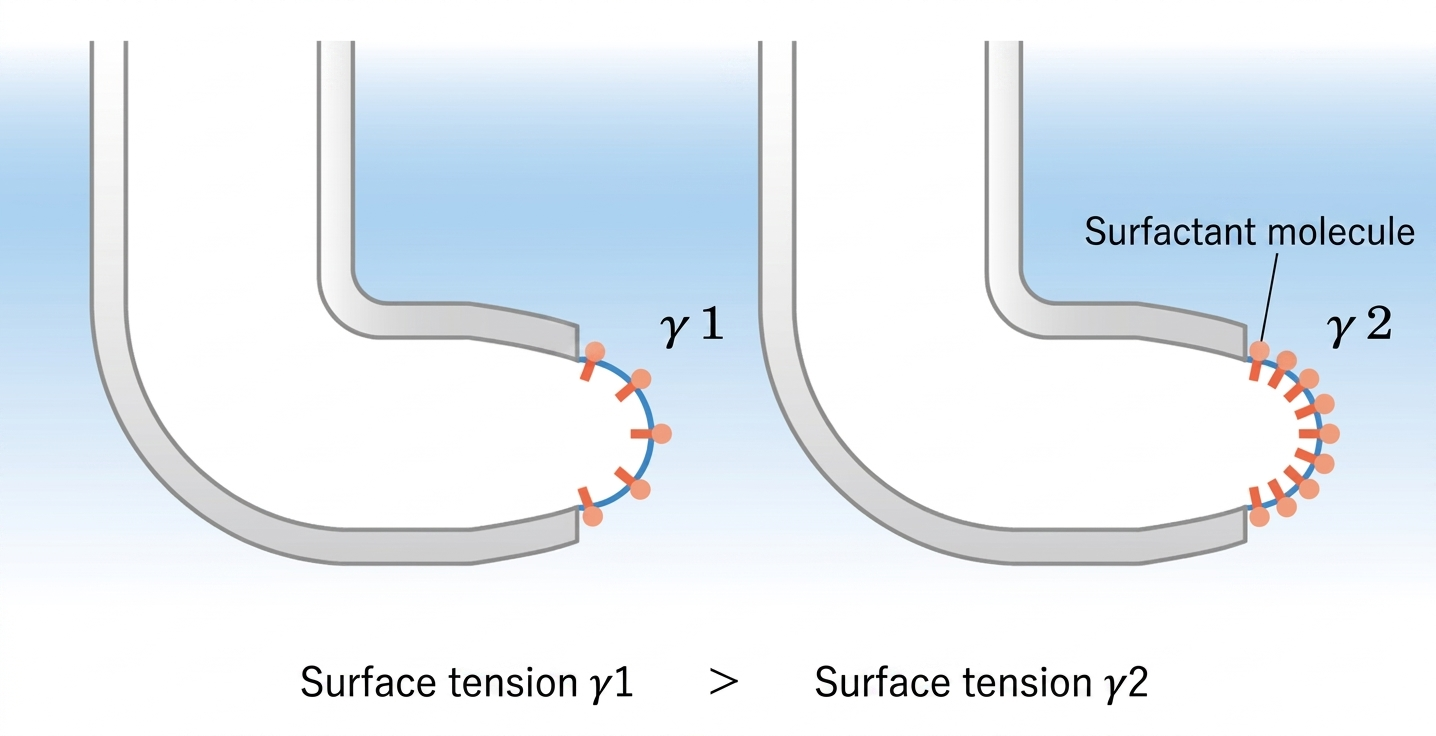
Using the Young-Laplace equation, the instrument determines the liquid's surface tension from the measured maximum differential pressure. During the bubble's lifetime, surfactant molecules continuously migrate from the bulk solution and adsorb onto the expanding gas-liquid interface. A longer lifetime allows more surfactants to accumulate at the surface, progressively lowering the dynamic surface tension until it approaches its static equilibrium value.









