Evaluation of surfactant properties of detergents using a dynamic surface tensiometer
Background
Introduction to the usefulness of dynamic surface tension measurements for evaluating surfactant adsorption behavior during the early stages of interface formation.
Problems to be solved
Traditional methods for measuring surface tension, such as the plate (Wilhelmy) and ring (du Noüy) methods, measure static surface tension, reflecting an equilibrium in which surfactants have fully adsorbed at the interface. Yet, in real-world situations such as cleaning, new interfaces constantly form due to agitation and flow, which prevents sufficient surfactant adsorption and often leads to higher readings than static values. Under these dynamic conditions, interfacial properties greatly influence wetting and foam formation. Nevertheless, relying solely on static surface tension measurements makes it difficult to fully understand interfacial behavior in practical settings. Thus, developing a measurement technique that evaluates adsorption during the initial stages of interface formation is crucial.
Surfactants lower surface tension by moving to and adsorbing at the air-liquid interface. Unlike static surface tension tests that measure equilibrium, dynamic tests can detect rapid changes within milliseconds right after the interface forms (see Figure 1). This article demonstrates the analysis of the interfacial properties of four commercial laundry detergents using our dynamic surface tensiometer.
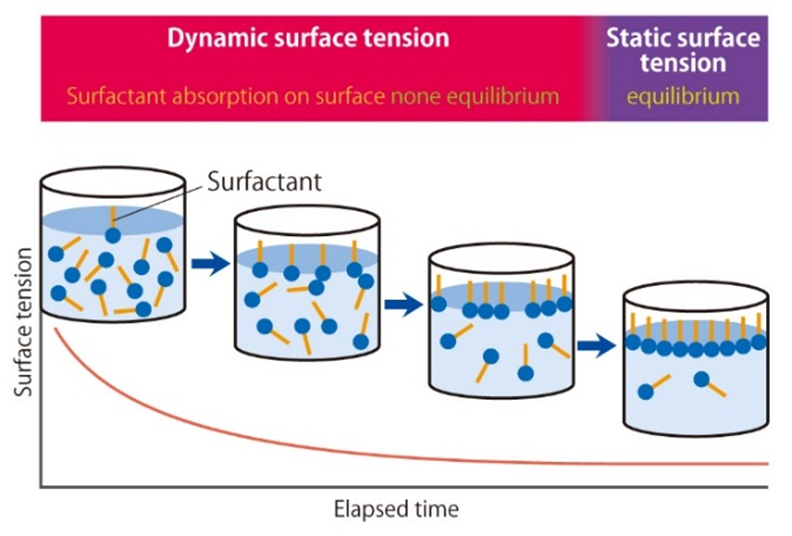
Figure 1 – Modelling surfactant adsorption over time
Measurements and results
Four laundry detergent product types were diluted to their respective use concentrations, and their static surface tension (measured via the plate method) and dynamic surface tension (measured via the maximum bubble pressure method) were assessed. Static surface tension was tracked over time and compared at 20 seconds, which was considered the point of equilibrium (see Figure 2). Product D exhibited the lowest static surface tension, whereas product B's value was the highest, suggesting a stronger surfactant effect at equilibrium. Throughout the measurement period, product B experienced the fastest drop in surface tension, indicating rapid adsorption at the interface. This resulted in a ranking that differed from the observations of static surface tensions.
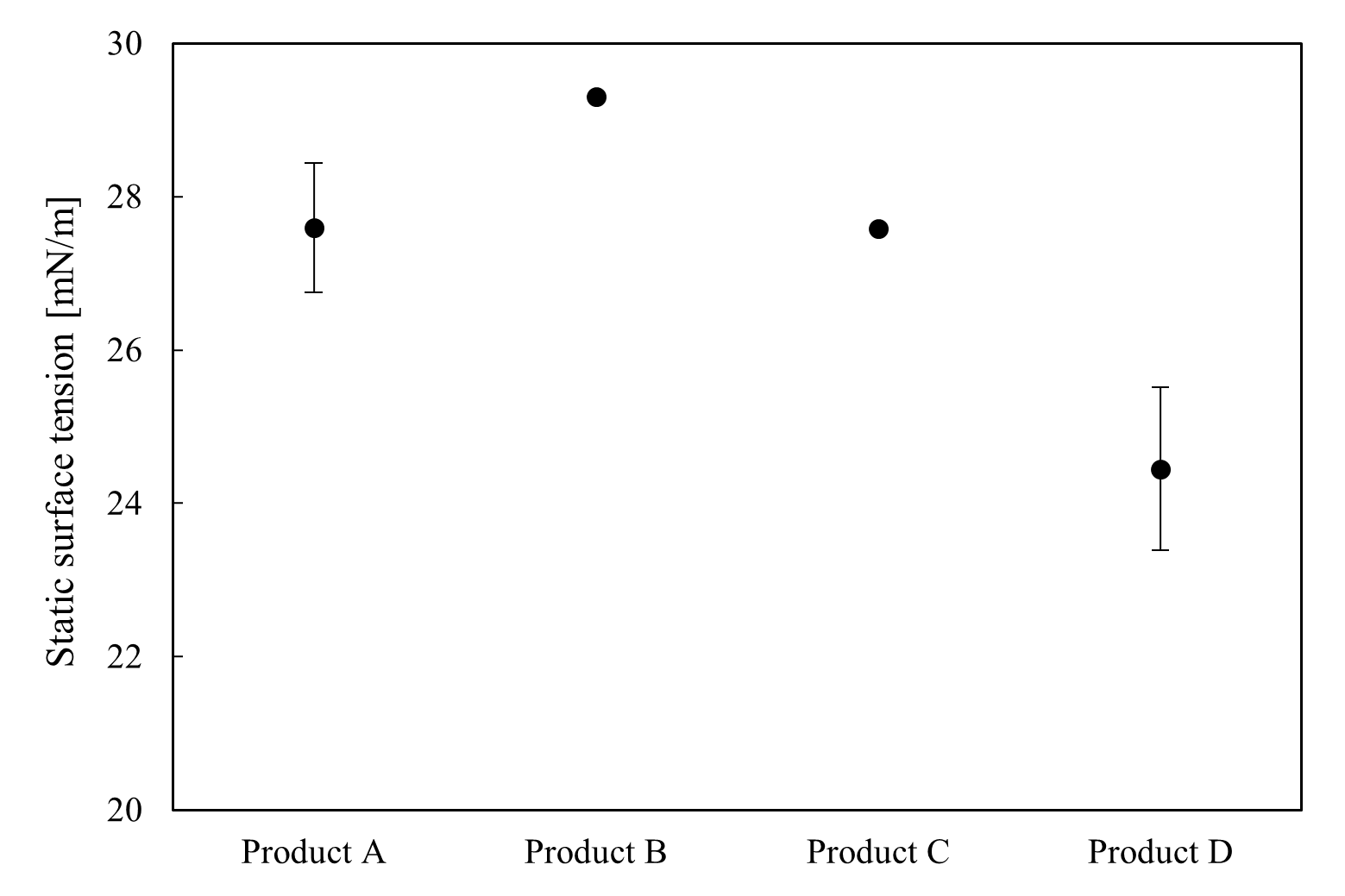
Figure 2 – Static surface tension results
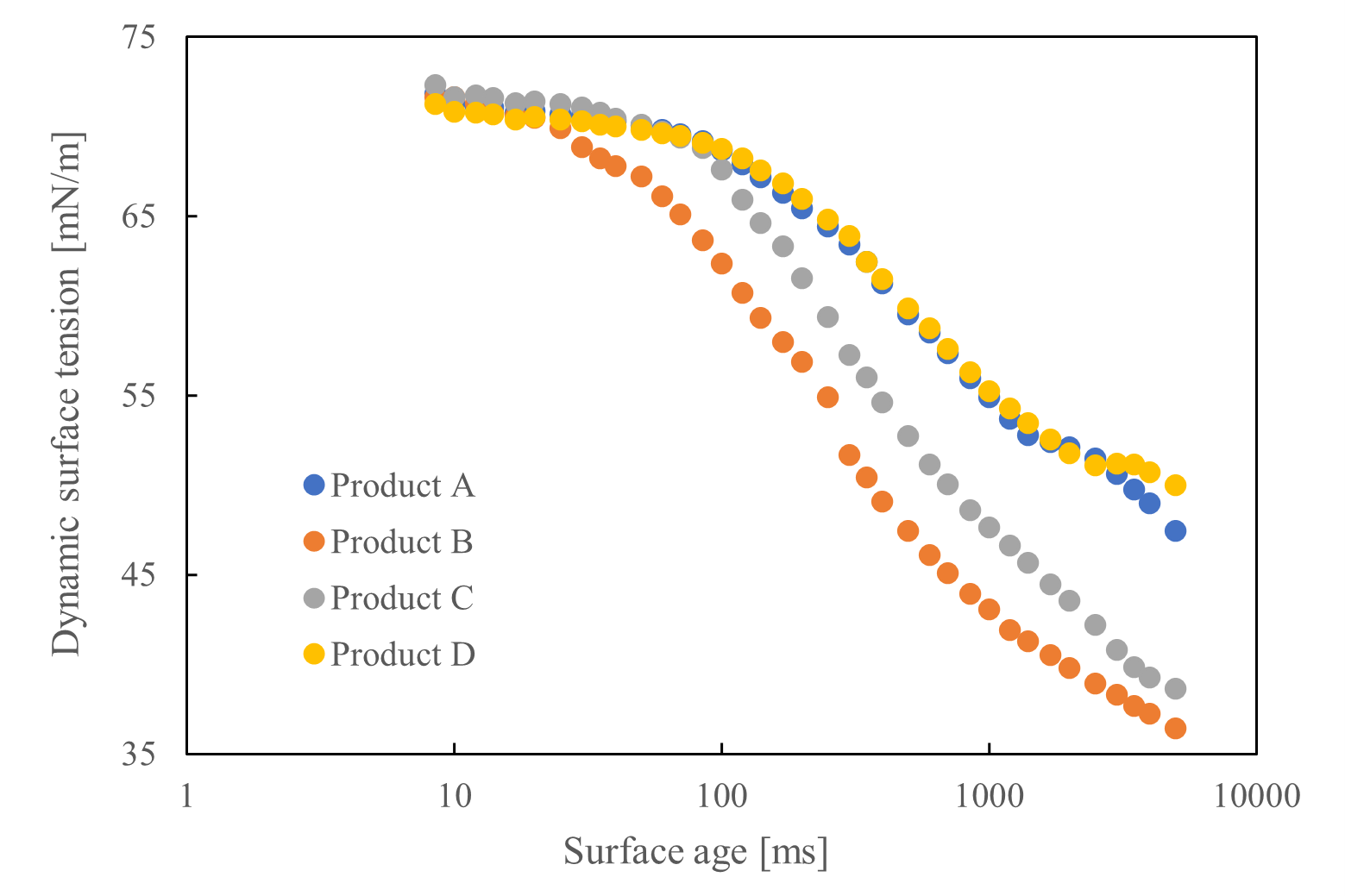
Figure 3 – Dynamic surface tension results
Conclusion
We verified that dynamic surface tension measurements can quantify surfactant adsorption behavior. Notably, the relationship between static and dynamic surface tension was opposite, underscoring the need for dynamic measurements alongside static ones when assessing products for dynamic conditions, such as detergents.







